A relatively high zoonotic trematode prevalence in Orientogalba ... - Infectious Diseases of Poverty - BioMed Central
Overall information on the sampling and survey data
Snail samples of O. ollula were collected from 54 sites (shown in Fig. 1 and Additional file 1: Fig. S1), with 38–214 snails from each site. Trematodes were found in 17 sites investigated following dissection, including Tianbao Reservoir and Hede village in Nanning city; Liushan Town, Liutang Village, Guangrong village, and Cha Village in Liuzhou City; and Maling Town in Guilin City. During the dissections, different developmental stages of the trematode (redia, cercaria and metacercariae) were found.

Detailed information related to the sampling sites, sample selection, and method of collecting information. A, B Typical ecological environment of O. ollula (Type 1 areas: rice cultivation areas); C O. ollula image in anterior view; D, E Typical ecological environment of O. ollula (Type 2 areas: vegetation areas of agricultural crops which often used as the raw food); F O. ollula image posterior view
Species identification of O. ollula
Morphological features of O. ollula were identified by traditional quantitative shell characteristics (shell: medium-sized to large, thin, colour brown or yellowish; shell height: 10 mm; shell width: 8 mm; four up to five separate brown spiral bands; aperture oval, slit-like umbilical foramen). O. ollula collected in this study were small, oval, and had a thin shell and a small aperture (Fig. 1C, F). The ITS1 data of all sequenced snails were 100% consistent with those of the previously reported sequences in GenBank under the accession number HM769888–HM769898.
Morphological character descriptions and molecular identification of trematode larvae
Generally, morphological characters of three echinostomes (Echinostoma sp., H. conoideum, and E. revolutum) showed considerable morphological similarity (Fig. 2). The rediae were cylindrical, blunt at both ends, slightly pointed at the head, and more pointed at the tail. The body was curved to the ventral surface with muscular feet, and the movement was slow. The tail of the cercariae was not forked (Fig. 2B–H). Beyond the general similarities, the head of H. conoideum cercariae showed prominent spines, as well as well-developed ventral suckers, pharynx, and intestines (Fig. 2D–F). The metacercariae were round and had two transparent walls, and the outer wall was thicker than the inner wall. Abdominal suckers and refractive granules of larvae could be seen inside the cyst. Due to the movement of the larvae inside the sac, the small spines around its head were not easily observed. The rediae of Australapatemon sp. forms a distinct bulge at the head. The cercaria larvae had a forked tail, which was longer than the body length. The cercaria of P. cordatum also had a visible forked-tail, oral sucker, and pharynx (Fig. 2J–L).
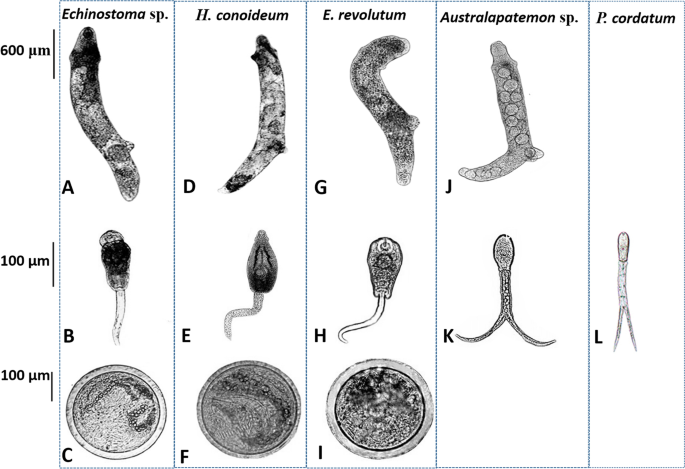
Morphology of rediae, cercariae, and metacercariae collected in O. ollula. A–C Rediae, cercariae and metacercariae of Echinostoma sp.; D–F Rediae, cercariae and metacercariae of Hypoderaeum conoideum; G–I Rediae, cercariae and metacercariae of Echinostoma revolutum; J, K Rediae and cercariae of Australapatemon sp.; L Cercariae of Pharyngostomum cordatum
The species of isolated trematodes were identified by amplification of the ITS2 region and verified through BLAST. BLAST analysis of sequences from sequenced trematode larvae showed the highest levels of sequence identity (99% to 100% sequence identity) to referenced sequences in GenBank. We identified five different species of trematodes, including Australapatemon sp., Echinostoma sp., E. revolutum, H. conoideum, and P. cordatum. The lengths of ITS2 were 292 bp, 429 bp, 430 bp, 432 bp, and 294 bp, respectively. The nucleotide sequences obtained in the present study have been deposited in the GenBank database under the accession numbers KM980466–KM980471 (Australapatemon sp.), KJ848453–KJ848455 (Echinostoma sp.), KM980474 and KM980476–KM980477 (E. revolutum), KJ944311–KJ944313 (H. conoideum) and OL870492 (P. cordatum).
Prevalence of trematodes in O. ollula
The overall trematode infection rate was 22.1% (1818/8238). E. revolutum was detected in the snails from 11 sampling sites, with an infection rate of 13.0% (1069/8238). Hypoderaum conoideum infection was detected in the snails from two sampling sites, with an average infection rate of 3.8% (315/8238). Australapatemon sp. was detected in the snails from 2 sampling sites, with an infection rate of 2.5% (206/8238). Infection of P. cordatum and Echinostoma sp. was detected at one sampling site with infection rates of 0.4% (34/8238) and 2.4% (194/8238), respectively. The overall prevalence and trematodes constituent ratio varied in type 1 and type 2 areas (P < 0.01).
Phylogenetic analyses
In total, 15 representative high-quality ITS2 sequence data were obtained. Figure 3 shows an NJ tree based on the submitted sequences and relevant GenBank sequences. The ITS2 sequences of Echinostoma sp. constituted a monophyletic clade (Fig. 3A shaded pink area), distinct from the clade formed by E. robustum, E. friedi, and E. miyagawai. The sequences of E. revolutum and H. conoideum constituted a monophyletic group together with E. revolutum (AY168930) and H. conoideum (AJ564385) references (Fig. 3A shaded blue area). The ITS2 sequences of Australapatemon sp. formed a group with A. burti (KU950451) at a 99% bootstrap value but formed a unique clade at 75% bootstrap value. Figure 3B shows that the ITS2 sequences of P. cordatum were identical to the reference sequences of P. cordatum (KJ137231).

Phylogenetic analyses of isolated trematodes based on the ITS2 sequences and relevant GenBank sequences. A Neighbor joining bootstrap consensus tree with 1000 bootstrap iterations for the rediae of echinostomes; B Neighbor joining bootstrap consensus tree with 1000 bootstrap iterations for Australapatemon sp. and Pharyngostomum cordatum
Laboratory infection experiment with Echinostoma sp., E. revolutum, and H. conoideum
We conducted an infection experiment for three kinds of trematode to evaluate the rates of parasite establishment in ducklings. Ducklings were individually exposed to Echinostoma sp. (n = 52), E. revolutum (n = 53), and H. conoideum larvae (n = 57), and all were successfully infected. During subsequent observation on ducklings (17 dpi) fed with Echinostoma sp. infected O. ollula, we detected eggs [(195.8 ± 3.1) × (143.8 ± 1.6) μm] in the feces, and the morphological characteristics of adult Echinostoma sp. were presented as measurements: body length (9.8 ± 1.8 mm), width (1.2 ± 2.3 mm), oral sucker [(638.9 ± 1.9) × (399.2 ± 15.8) μm], acetabulum [(1591.2 ± 17.8) × (1338.2 ± 19.6) μm], pharynx [(492.6 ± 54.3) × (331.7 ± 74.8) μm], anterior testis [(1120.5 ± 139.6) × (707.4 ± 49.2) μm], posterior testis [(1274.9 ± 181.1) × (880.4 ± 49.2) μm], and ovary [(818.9 ± 10.2) × (527.9 ± 48.3) μm].
In contrast, we found H. conoideum eggs in ducklings fed with infected O. ollula from three sites after a median duration of 12 dpi (range: 9 dpi to 14 dpi). The morphological characteristics of adult H. conoideum were as follows: body length (1.05 ± 0.61 mm), width (1.5 ± 0.27 mm), oral sucker [(424.2 ± 16.1) × (293 ± 11.2) μm], acetabulum [(1610.2 ± 98.4) × (1594.6 ± 198.7) μm], pharynx [(379.8 ± 53.2) × (253.6 ± 44.9) μm], anterior testis [(1902.6 ± 164.6) × (875.3 ± 140.8) μm], posterior testis [(2045.2 ± 255.8) × (898.2 ± 139.2) μm], ovary [(751.7 ± 129.3) × (553.9 ± 110.6) μm], and the possession of 50 spines. E. revolutum eggs [(104.1 ± 15.1) × (63.1 ± 12.6 μm] were found on 10 dpi. The morphology of the adult E. revolutum was characterized by: body length (8.5 ± 1.2 mm), width (2.2 ± 0.7 mm), oral sucker [(260.0 ± 32.5) × (180.1 ± 19.8) μm], acetabulum [(741.6 ± 23.7) × (598.3 ± 25.1) μm], pharynx [(193.1 ± 30.5) × (150.6 ± 27.8) μm], anterior testis [(628.0 ± 31.2) × (459.4 ± 27.8) μm], posterior testis (725.5 ± 17.1 × 557.9 ± 18.7 μm), ovary [(411.8 ± 18.5) × (311.2 ± 15.4) μm], and the presence of a head collar with 37 spines.
Developmental characteristics of E. revolutum, from juvenile to adult, in duckling hosts
Experiments were only designed to study the development of E. revolutum in duckling hosts, because there were insufficient snails infected with metacercariae of Echinostoma sp. and H. conoideum. The developmental characteristics of E. revolutum were recorded by dissecting infected ducklings from 1 to 10 dpi when eggs in the feces were first detected. E. revolutum could be obtained in the small intestine from 1 to 7 dpi and then migrate and reside in the cecum and colon from 8 to 10 dpi. The body length developed from 490 μm to 8500.5 μm (a 17-fold increase). At 1 dpi, juveniles had a circumoral collar bearing 37 spines in a double circle and characterized by clearly visible oral suckers, acetabulum, pharynx, esophagus, and cecum. At 1 dpi, the tiny structure of the testis appeared. By 4 dpi, the ovaries were beginning to organize and develop, and the seminal receptacle began to form. The tubular-shaped uterus loomed at 4 dpi, and maturation of the reproductive and digestive organs occurred at 6 to 8 dpi. The vitelline glands were the last to appear, and several eggs deposited in the uterus were observed at 9 dpi. E. revolutum larvae matured at 10 dpi and excreted eggs (Fig. 4). The daily development of E. revolutum development was recorded in detail, and the results are shown in Additional file 3: Table S2.

Development of E. revolutum in duckling host from 1 to 10 dpi
Comments
Post a Comment